Upneeq: Caffeine for your Eyes
Upneeq® is an innovative eye drop that acts like a coffee boost for your eyes! Bye-bye, droopy lids, and welcome, brighter, more awake eyes. Upneeq® is the first and only remedy for acquired ptosis, gently raising your upper eyelids to restore your confidence in seeing the world. We are honored to be your dependable provider for this eye-opening transformation.
Upneeq® is where your vivid, wide-eyed look begins!


Upneeq delivered to your door!
We take great satisfaction in being your trusted Upneeq eye drops provider, as we are at the forefront of eye care innovation. Upneeq® is the thrilling breakthrough that’s making waves in the world of eye care, and we’ve got the secret to brighter, more vibrant eyes. Say goodbye to acquired ptosis (eyelid drooping) as Upneeq® enters the picture, providing a safe and effective remedy. Our method is seamless and transformative: Begin by completing our free medical history form, then let our expert eye care professionals take it from there.
Following a thorough review, we will promptly offer you a unique email and text link connecting you directly to RVL, the manufacturer of Upneeq®. You can then easily place your order, and your Upneeq® products will be delivered to your house.
We Gave The
Best For You
Delivered To Your Door
With its cleverly packed 30-pack, Upneeq redefines convenience, a testament to the future of eye care. Each individually wrapped unit is sterile and preservative-free, ensuring your Upneeq® experience is as pleasant and pure as possible.Upneeq® believes in improving your well-being at every step of the road. That's why our upneeq samples are designed to be delivered right to your door by the expert Upneeq provider, saving you time and effort
The Science Stuff
Upneeq contains active chemicals that interact with the musculature of your eyes, elevating the upper eyelids effectively.The science behind Upneeq® is based on its capacity to increase muscle contraction and improve the function of the levator palpebrae superioris muscle, providing you with the gift of wide-open, vivid eyes. Upneeq samples promote the release of norepinephrine, a neurotransmitter that enhances muscle activity, by activating the sympathetic nervous system.
How do I use it?
Upneeq, the modern eye care marvel, digs into the exciting world of scientific innovation. This ground-breaking eye solution is carefully developed to treat acquired ptosis or drooping eyelids. Eye care becomes a breeze with Upneeq®. Apply one drop to each eye once a day, as directed. Whether you use it daily or for special occasions is up to you, but consult your eye specialist for tailored guidance on maximizing Upneeq while safeguarding your eye health.
Your Vision, Our Priority
Upneeq values your vision, so we want you to know the risks and warnings associated with our revolutionary solution.
Upneeq is a fantastic medication developed to revitalize your eyes by treating acquired ptosis, but it has drawbacks. It is critical to understand that Upneeq has the potential to affect your blood pressure. If you have heart disease or uncontrolled high or low blood pressure, use Upneeq cautiously. If you ever suffer dizziness or light-headedness, especially when moving from sitting or lying down to standing, it’s critical to contact your doctor if these symptoms worsen.Your eye care journey is our joint commitment at Upneeq. We encourage you to prioritize your health and seek medical attention if needed. Everything we do is focused on your eyesight, comfort, and safety

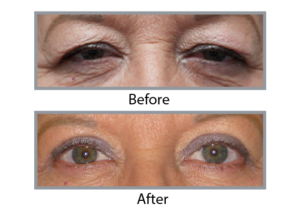
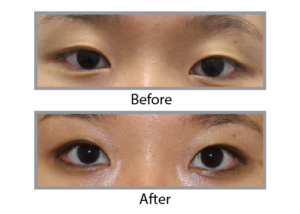
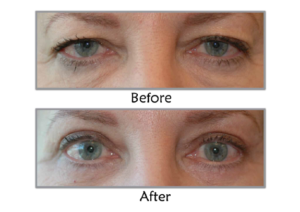
Real Stories, Real Results
FAQs: Your Upneeq Queries
A box of 30 packs of Upneeq currently is $160, but down to $116 on a subscription plan (cancel anytime)
Yes, Upneeq eye drops are only available with a prescription from a competent eye doctor. Upneeq eye drops treat acquired ptosis characterized by drooping eyelids. They should be taken under the supervision and guidance of a healthcare expert because they contain active medication.
To receive Upneeq, you must first consult with an eye doctor provider who will analyze your condition and, if necessary, prescribe the prescription. It is critical to follow their instructions for safe and effective use.
Upneeq is a prescription eye drop used to treat acquired ptosis or drooping upper eyelids. It works by temporarly stimulating and contracting the Müller’s muscle, which lifts the upper eyelid and improves the aesthetic appearance and field of vision.
This innovative solution offers a temporary but effective way to combat droopy eyelids, making your eyes appear more alert and rejuvenated. Always use Upneeq as your healthcare provider directs for the best results to ensure safety and the desired cosmetic benefits.
Upneeq is generally considered safe for daily use as directed by a healthcare professional. One drop in each eye, once a day, is the usual suggested dosage.
They will examine your unique condition, medical history, and potential adverse effects to select the most appropriate use. Upneeq does not have to be used every day to work effectively.
For personalized suggestions, always consult with your healthcare professional.
Upneeq’s effects were seen to peak around 1-2 hours after instillation and could extend for up to 8-10 hours in clinical trials.
Individual circumstances can influence the length, so it’s critical to follow your healthcare provider’s recommendations for when and how often to use Upneeq, depending on your needs and response to the drug.
If you have questions concerning the length of Upneeq’s effects, talk to your eye doctor or prescribing healthcare practitioner.
Upneeq can temporarily improve the appearance of drooping or ptotic upper eyelids, making some people appear more alert and rejuvenated. As a result, it may provide a cosmetic benefit associated with a more youthful appearance, which some people associate with.
However, it’s important to note that Upneeq has no long-term anti-aging effects on the skin or the underlying structures of the eye. The cosmetic improvement achieved with Upneeq is temporary and reversible, typically lasting several hours after application.
Upneeq should not be used more than once per day unless specifically directed by your healthcare provider. Upneeq is usually prescribed once a day, with one drop in each eye.
Using Upneeq more frequently than prescribed can increase the risk of potential side effects or reduced effectiveness. Following your doctor’s recommendations regarding the dosing regimen for your specific condition is essential.
If you have concerns or questions about your Upneeq usage, it’s best to discuss them with your healthcare provider for personalized guidance.
Beverly Hills Eye Care is a reliable Upneeq provider. Fill out the form accurately. After our doctor has reviewed it, you will be issued a link through email and SMS to place your order directly with the manufacturer, RVL.
REACH OUT TO US WITH
ANY QUESTIONS


